Abstract
Background: In utero hematopoietic cell transplantation (IUHCT) results in long-term, multilineage chimerism without myeloablation/immunosuppression. It has the potential to treat a number of congenital hematologic disorders, including as Sickle cell disease. Central tolerance, through which donor-reactive host T cells and host-reactive donor T cells are deleted, is instrumental to the induction of donor-specific tolerance (DST) and the prevention of graft-versus-host-disease (GVHD). The window of central tolerance induction in humans occurs at a gestational age that will pose significant technical challenges and exclude from treatment all fetuses evaluated after 14 weeks gestation. We have previously demonstrated that IUHCT results in the peripheral induction of traditional and non-traditional regulatory T cells that contribute to the suppression of GVHD (Riley et al. ASH 2016 Abstract #4540). We hypothesized that regulatory T cells also contribute to DST and, furthermore, may be used to expand the window of central tolerance induction to a later point in gestation.
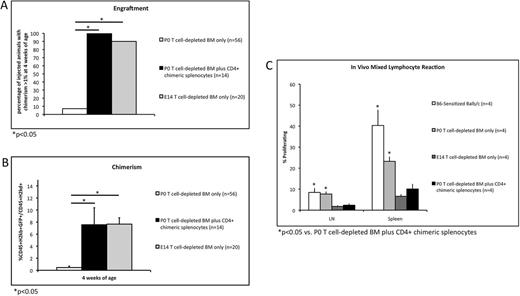
Methods: A mouse model of delayed gestational age IUHCT was developed in which 9.7x10^6 C57/Bl6 (B6) GFP T cell-depleted (TCD) bone marrow (BM) cells were injected intravenously into day of life 0 (P0) naive Balb/c pups via the facial vein. The ability of IUHCT-induced regulatory T cells to prevent donor cell rejection was assessed by injecting 9.7x10^6 B6 GFP TCD BM cells plus 5x10^6 CD4+ splenocytes harvested from 2-4 week old chimeric mice. Fetuses injected with 9.7x10^6 B6 GFP TCD BM cells at gestational day 14 (E14) served as positive controls for tolerance and engraftment. Peripheral blood (PB) was analyzed at 4, 8, 12, and 24 weeks of age for donor cell chimerism (%CD45+H2kb+GFP+/CD45+H2kd+) and for engraftment (defined as chimerism >1% at 4 weeks). Weight gain and histology of the skin, liver, lungs, and small bowel were analyzed to confirm the absence of GVHD. PB was analyzed at 6 months of age to confirm multilineage engraftment. An in vivo mixed lymphocyte reaction (MLR) was performed at 6 months of age to confirm long-term DST. B6-sensitized Balb/c mice served as positive controls for alloreactivity. Comparison of continuous data was performed using Student's t test. Binary data were compared using Fischer's exact test. All tests were two-sided with an alpha level set at 0.05 for significance.
Results: Our model of delayed gestational age IUHCT was associated with donor cell rejection in 52 of 56 (92.9%) pups injected with TCD BM only. Co-injection of CD4+ chimeric splenocytes significantly improved engraftment (100% vs. 7.1%, p<0.001) and chimerism (7.56% vs. 0.46% at 4 weeks, p=0.003) compared to TCD BM alone. No difference in engraftment (100% vs. 90%, p=0.501) or chimerism (7.56% vs. 7.66% at 4 weeks, p=0.961) was observed compared to fetuses injected with TCD BM at E14 (Figures 1A and 1B). These trends persisted at subsequent time points. Pups injected with TCD BM plus CD4+ chimeric splenocytes displayed normal weight gain and no histologic evidence of GVHD. Survival to adulthood was 100%. Donor cells of all hematopoietic lineages were present in the PB at 6 months of age. On in vivo MLR, pups injected with TCD BM plus CD4+ chimeric splenocytes displayed significantly reduced T cell proliferation compared to B6-sensitized Balb/c mice and P0 pups injected with TCD BM only. T cell proliferation did not differ significantly from fetuses injected with TCD BM at E14 (Figure 1C).
Conclusion: The induction of tolerance following IUHCT is important for maintaining successful long-term engraftment and requires injection at an early gestational age to avoid donor cell rejection by the maturing fetal immune system. This study demonstrates an important role for regulatory T cell populations in the induction of DST following IUHCT. The adoptive transfer of regulatory T cells from a chimeric mouse to a naïve P0 pup inhibits donor cell rejection in our model of delayed gestational age IUHCT, allowing central tolerance induction and long-term engraftment to occur despite the increased maturity of the recipient immune system. These findings represent proof-of-principle that augmentation of regulatory T cell-dependent peripheral tolerance is one approach by which IUHCT could be delayed to a later point in gestation, decreasing the technical difficulty of the procedure and allowing fetuses evaluated after 14 weeks to be eligible for treatment.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal